Equipment and systems qualification is a critical element in ensuring product quality, regulatory compliance, and patient safety in the pharmaceutical industry. In this context, the Factory Acceptance Test (FAT) and the Site Acceptance Test (SAT) are decisive, as they allow for structured and documented verification that equipment meets technical, functional, and regulatory requirements before going into operation.
In an increasingly demanding environment, marked by rigorous regulatory inspections, highly automated systems, and a growing focus on digitalization and sustainability, FAT and SAT have evolved significantly. They are no longer viewed merely as technical tests but are now fully integrated into the GMP (Good Manufacturing Practice) validation cycle and the regulatory preparation of organizations.
By identifying and addressing deviations at an early stage, FAT and SAT help minimize technical and regulatory risks, ensuring a more controlled and predictable transition between equipment installation, start-up, and ramp-up phases. This approach facilitates handover to operations teams, reduces instability during initial production cycles, and ensures systems become operational safely and efficiently, laying the foundation for operational excellence throughout the asset lifecycle.
This article explores the roles of FAT and SAT in the pharmaceutical industry, from their place in the GMP validation cycle to best practices, key challenges, and future trends.
Understanding FAT and SAT in the pharmaceutical context
In the pharmaceutical industry, the Factory Acceptance Test and the Site Acceptance Test are fundamental stages in the equipment and systems qualification lifecycle. These tests provide a structured and documented approach to verifying that production equipment, critical systems, and laboratory instruments meet technical, functional, and regulatory requirements before being integrated into routine operations.
What Is the Factory Acceptance Test (FAT)?
The Factory Acceptance Test (FAT) is performed at the supplier’s facility before the equipment is shipped to its final installation site. Its main objective is to confirm that the equipment or system has been built in accordance with the approved User Requirements Specifications (URS) and technical specifications.
In the pharmaceutical context, the FAT typically includes:
- Verification of the equipment’s functionality and its critical components.
- Automation tests, alarms, emergency stop buttons, and operational sequences.
- Preliminary evaluation of GMP requirements, including data integrity and safety aspects.
- Review of technical documentation, such as electrical schematics, component lists, and software versions.
Effective execution of the FAT allows for early identification and correction of deviations, reducing costs, delays, and risks associated with late-stage changes after installation. Furthermore, the FAT provides a solid foundation for the subsequent qualification phases.
What Is the Site Acceptance Test (SAT)?
The Site Acceptance Test (SAT) is conducted after the equipment has been installed at the pharmaceutical manufacturer’s site. The SAT focuses on verifying that the equipment functions properly in its actual operating environment, taking into account interfaces, utilities, layout, flows, and support systems.
In the pharmaceutical sector, the SAT typically involves:
- Confirmation of proper installation and connection to utilities (power, compressed air, water, HVAC, among others).
- Functional testing of the equipment integrated within the production or laboratory environment.
- Verification of communication with automation and data management systems, such as MES, SCADA, or LIMS.
- Assessment of on-site compliance with safety, ergonomics, and GMP requirements.
The SAT ensures that the equipment is ready to proceed to formal qualification, reducing uncertainties and confirming that the performance observed during FAT is replicated under real operating conditions.
Key differences between FAT and SAT
Although complementary, FAT and SAT serve different purposes and are performed in distinct contexts. FAT focuses on equipment compliance with technical specifications before shipment, while SAT validates equipment performance after installation at the final site.
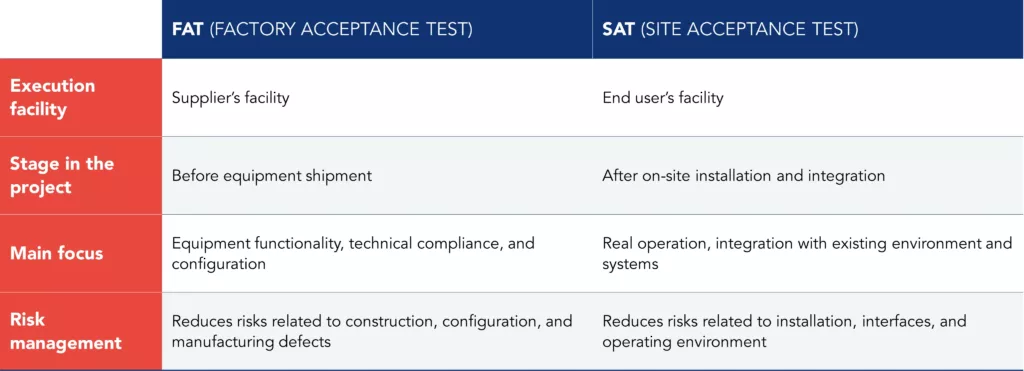
Table 1 – Key differences between FAT and SAT in the pharmaceutical industry
Together, FAT and SAT testing constitute a structured approach that is essential for ensuring technical robustness, regulatory compliance, and improved operational efficiency in the pharmaceutical industry.
Reduce lead time and gain operational predictability with well-structured FAT and SAT
FAT and SAT as the foundation of (Good Manufacturing Practice) validation and regulatory preparedness
In the pharmaceutical industry, GMP validation requires that all critical equipment, systems, and processes are demonstrably fit for their intended use before the start of commercial production. In this context, the Factory Acceptance Test and the Site Acceptance Test play a foundational role, serving as essential control points throughout the validation lifecycle.
The role of FAT and SAT in the GMP validation cycle
FAT and SAT take place in the early phases of the GMP validation cycle, formally preceding the Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ) stages. FAT verifies at the supplier’s site whether the equipment meets the requirements defined in the URS and technical specifications, while SAT confirms that these requirements are maintained after installation at the final site.
This approach:
- Supports the lifecycle-based validation model.
- Enables early identification and mitigation of technical and functional risks.
- Ensures alignment between initial requirements and final system performance.
When well-planned, FAT and SAT reduce the burden of repetitive testing during qualification stages and facilitate the transition to formal validation in accordance with GMP principles.
Alignment with international standards
FAT and SAT must be planned and executed in alignment with the main regulatory and industry standards in the pharmaceutical sector. Key references include:
- EU GMP Annex 15, which establishes the principles of qualification and validation.
- FDA 21 CFR Part 11, regarding computerized systems and data integrity.
- ICH Q7, applicable to active pharmaceutical ingredients (APIs).
- GAMP 5, as a guide for validating automated systems.
Alignment with these standards ensures that FAT and SAT outcomes are accepted by regulatory authorities, reducing the risk of critical findings during inspections and audits.
Ensuring traceability, data integrity, and audit readiness
One of the most significant contributions of FAT and SAT to GMP validation is the generation of traceable and verifiable documentary evidence. Well-structured protocols allow each test to be linked directly to URS requirements, ensuring a clear traceability chain.
Additionally, the increasing use of electronic records, digital signatures, and automated systems during FAT and SAT enhances data integrity, ensuring that information is complete, consistent, accurate, and protected against unauthorized changes.
This approach facilitates preparation for both internal and external audits, enables rapid response to evidence requests from regulatory authorities, and provides objective proof of GMP compliance and control.
Reducing validation risks and accelerating qualification timelines
Identifying and resolving deviations during FAT and SAT has a direct impact on reducing validation risks. Issues detected early are generally simpler and less costly to address than those identified after the start of formal qualification or production.
Moreover, when FAT and SAT are well executed:
- They reduce rework during IQ and OQ.
- They minimize delays in equipment start-up.
- They support on-time project delivery and product launch schedules.
In this way, FAT and SAT become accelerators of the qualification process, ensuring regulatory compliance, operational efficiency, and project predictability in pharmaceutical environments.
Digital transformation and sustainability in pharmaceutical equipment qualification
The pharmaceutical industry’s shift toward more digital and sustainable models is transforming how equipment is qualified. In this new context, the Factory Acceptance Test and Site Acceptance Test are no longer just technical verification checkpoints; they now play a key role in digital transformation and in integrating sustainability and energy efficiency criteria from the early stages of the asset lifecycle.
Incorporating these new drivers into qualification not only reinforces GMP compliance but also improves operational efficiency, data transparency, and alignment with organizations’ environmental and strategic goals.
FAT and SAT as catalysts of digital transformation (Pharma 4.0)
Within the context of Pharma 4.0 and the growing adoption of AI in the pharmaceutical industry, FAT and SAT are evolving into increasingly digital, automated, and integrated processes. The use of digital FAT and SAT, leveraging collaborative platforms, enables remote testing and virtual acceptance, reducing travel, shortening timelines, and facilitating the involvement of geographically distributed multidisciplinary teams.
The application of digital twins and automated test protocols allows simulation of equipment behavior prior to installation, enhancing performance predictability and test robustness. These digital models enable early and structured validation of control logic, operational sequences, and failure scenarios.
Additionally, integration of tested equipment with systems such as MES, SCADA, and LIMS is increasingly validated during FAT and SAT, ensuring connectivity, consistent data flows, and compliance with data-integrity requirements. Real-time deviation management, combined with electronic documentation and auditable records, strengthens traceability and simplifies preparation for regulatory audits.
Integrating sustainability and energy efficiency into FAT and SAT
Sustainability is becoming an essential dimension of pharmaceutical equipment qualification. FAT and SAT increasingly include the evaluation of environmental and energy efficiency criteria in addition to traditional functional and safety requirements.
During these phases, indicators such as the following are analyzed:
- Energy consumption of the equipment and associated systems.
- Ventilation and HVAC system efficiency, which is especially critical in cleanroom environments.
- CO₂ emissions and environmental impact throughout the equipment’s lifecycle.
Integrating these parameters enables informed decision-making before the equipment enters operation, helping avoid energy-inefficient solutions or those misaligned with the organization’s environmental commitments. At the same time, it ensures alignment between GMP requirements and corporate ESG objectives, promoting facilities that are more responsible, efficient, and future-ready.
Best practices for effective FAT and SAT execution in the pharmaceutical industry
The effectiveness of FAT and SAT depends less on the number of tests performed and more on the quality of planning, the clarity of acceptance criteria, and the robustness of execution. In the pharmaceutical industry, where regulatory requirements are stringent, adopting structured best practices is essential to ensure GMP compliance, operational efficiency, and project predictability.
Early planning with a risk-based approach
Effective FAT and SAT begin with early planning, aligned with a risk-based approach as outlined in GMP guidelines. This approach allows teams to focus efforts on the systems, functions, and parameters with the greatest impact on product quality, patient safety, and data integrity.
Planning should include:
- Identification of critical systems and critical functions.
- Clear definition of FAT and SAT scope based on risk.
- Alignment between URS, functional specifications, and test protocols.
- Timely involvement of engineering, quality, IT, and operations teams.
This alignment reduces ambiguity, avoids test duplication, and ensures a smooth transition to the formal qualification phases.
Collaboration with suppliers and clear definition of acceptance criteria
Close collaboration with suppliers is a critical success factor in FAT and SAT execution. From the early stages of the project, it is essential to ensure that acceptance criteria are clearly defined, documented, and understood by all stakeholders.
Best practices include:
- Defining objective, measurable acceptance criteria aligned with the URS.
- Joint review of FAT and SAT protocols.
- Clarifying responsibilities for deviation resolution.
- Involving the supplier in preparing technical and validation documentation.
A transparent and collaborative relationship helps with early problem detection and enables quicker and more effective resolution.
Deviation management and change control during FAT and SAT
Deviations during FAT and SAT are to be expected, especially in complex or highly automated projects. The key differentiator lies in how those deviations are identified, assessed, and resolved.
Effective deviation management should include:
- Structured and traceable recording of deviations.
- Impact assessment on quality, safety, and GMP compliance.
- Clear definition of corrective actions and associated responsibilities.
- Strict application of change control processes.
Disciplined deviation management during FAT and SAT prevents issues from spreading to later stages, reduces validation risks, and ensures a more robust and predictable qualification process.
Adopting best practices in FAT and SAT execution not only ensures GMP compliance but also lays a strong foundation for continuous improvement in the pharmaceutical sector by enabling the systematic incorporation of lessons learned into future projects.
Key challenges and how to overcome them
Although well-established practices in the pharmaceutical industry, the execution of FAT and SAT continues to present significant challenges, especially in a context of increasing technological complexity, heightened regulatory demands, and tighter project timelines. Identifying these challenges and adopting structured approaches to overcome them is essential to ensure the effectiveness of acceptance testing and the overall success of the qualification process.
Incomplete requirements and late design changes
One of the most frequent challenges in executing FAT and SAT involves incomplete, ambiguous, or poorly defined requirements during the early phases of a project. Vague URS or misalignment with the process’s actual needs often lead to late-stage design changes, directly impacting acceptance testing.
To mitigate this risk, it is critical to:
- Invest time in a thorough definition and review of the URS.
- Involve key areas early on (engineering, quality, operations).
- Apply a risk-based approach to prioritize critical requirements.
- Formalize changes through established change control processes.
These practices help reduce rework, delays, and potential deviations during FAT and SAT.
Gaps in data integrity and documentation
The increasing digitalization of pharmaceutical equipment and systems raises the risk of data integrity gaps, particularly when FAT and SAT processes do not adequately address regulatory requirements for computerized systems.
Common issues include:
- Incomplete or inconsistent records.
- Lack of traceability between requirements, tests, and results.
- Inadequate access controls and audit trails.
- Poor version management and approval tracking.
Adopting structured protocols, validated electronic documentation, and data integrity principles (ALCOA+) is essential to ensure GMP compliance and audit readiness.
Coordination between engineering, quality, and IT
Effective execution of FAT and SAT requires close collaboration between Engineering, Quality, and IT, something that doesn’t always occur seamlessly. Communication breakdowns or poorly defined responsibilities can result in test gaps, delays in decision-making, and conflicts during deviation resolution.
To overcome this challenge, it is recommended to:
- Clearly define roles and responsibilities.
- Establish regular alignment and decision-making mechanisms.
- Promote an integrated approach throughout the project lifecycle.
- Involve IT from the earliest stages, especially for automated and integrated systems.
It is also important to recognize that many challenges associated with FAT and SAT execution are not purely technical or regulatory in nature. Often, they stem from gaps in leadership, risk-based decision-making, and coordination among multidisciplinary teams. Developing leadership focused on managing complex projects is, therefore, a critical factor in ensuring cross-functional alignment, accelerating deviation resolution, and securing the overall robustness of the qualification process.
Learn how to ensure compliance and efficiency in the pharmaceutical industry
The future of FAT and SAT in the pharmaceutical industry
The role of FAT and SAT will continue to evolve as the pharmaceutical industry faces new regulatory, technological, and environmental challenges. The future of these acceptance tests points toward an increasingly digital, integrated, risk-based, and lifecycle-oriented approach, reinforcing their contribution to GMP compliance, operational efficiency, and organizational resilience.
In the coming years, it is expected that:
- Digitalization and remote acceptance models will become standard practices, supported by collaborative platforms and validated electronic evidence.
- Early integration with digital systems (MES, SCADA, LIMS, and analytics platforms) will be systematically validated during FAT.
- Data analysis and automated testing will provide greater performance predictability and earlier fault detection.
- Sustainability and energy efficiency criteria will be consistently incorporated into acceptance protocols.
- Regulatory authorities will heighten expectations around traceability, data integrity, and lifecycle visibility of equipment.
In this context, FAT and SAT will no longer be viewed merely as formal acceptance steps, but will become pillars of regulatory readiness, digital transformation, and sustainability in the pharmaceutical industry.
Conclusion: From acceptance testing to strategic advantage
Factory Acceptance Tests and Site Acceptance Tests have moved beyond being seen as purely technical verification steps to become key structural elements of GMP validation, regulatory preparedness, and operational efficiency in the pharmaceutical industry.
When rigorously planned and executed, FAT and SAT enable early risk mitigation, ensure compliance from the initial stages of a project, and significantly reduce the effort and uncertainty in subsequent qualification and validation phases. Their integration with digitalization, data management, and sustainability practices further enhances their value, turning them into catalysts for more agile, transparent, and responsible operations.
In a landscape marked by increasingly complex systems, growing regulatory demands, and pressure to accelerate time-to-market, organizations that treat FAT and SAT as strategic tools for risk management, process optimization, and sustainable value creation will be better equipped to meet regulatory expectations, optimize investments, and build pharmaceutical facilities truly ready for the future.
Still have some questions about FAT and SAT in the pharmaceutical industry?
What is GMP in the pharmaceutical industry?
GMP (Good Manufacturing Practice) is the set of principles, standards, and regulatory requirements that ensure medicines are consistently produced, controlled, and released with the necessary quality, safety, and efficacy for the patient.
In the pharmaceutical industry, GMP covers all critical areas, including facilities, equipment, processes, quality control, documentation, data integrity, and qualification and validation. It is a mandatory requirement for compliance with regulatory authorities.
How do FAT and SAT apply to quality control in the pharmaceutical industry?
Factory Acceptance Tests (FAT) and Site Acceptance Tests (SAT) apply to the equipment and systems used in pharmaceutical quality control, serving as key steps in the validation of quality control laboratories. These tests verify the functional compliance of analytical equipment, the fulfillment of User Requirements Specifications (URS), and GMP (Good Manufacturing Practice) requirements, creating a solid foundation for Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ), while reducing the risk of analytical deviations and batch release delays.
How do FAT and SAT contribute to operational efficiency and continuous improvement in the pharmaceutical sector?
By identifying deviations at an early stage, FAT and SAT reduce rework, delays, and instability during equipment start-up. This approach lays the groundwork for operational efficiency and continuous improvement in the pharmaceutical sector, enabling process optimization, the incorporation of lessons learned into future projects, and the overall enhancement of organizational maturity in terms of quality and performance.
See more on Pharmaceuticals
Find out more about transformation in this sector
